- Blog
- Turnitin uf
- Obd-ii j2534 elm327 toad diagnostic software
- Dodonpachi resurrection black label xbox marketplace
- Contract killer 2 mod apk revdl
- Autodesk 3ds max 2012 material library download
- How to do edge blending in propresenter 6
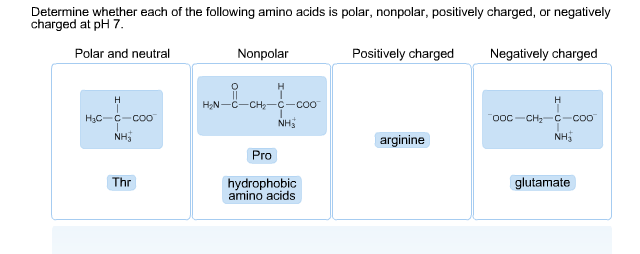
- Are hydrophobic amino acids nonpolar
- Alien shooter pc
- Visual studio enterprise subscription azure
- Contoh proposal manajemen keuangan
- Clipp store download
- Regulador voltaje o supresor de picos
- How to add subtitles to video file permanently
J Chem Soc Perkin Trans 2: 79–84Įl Tayar N, Testa B, Carrupt PA (1992b) Polar intermolecular interactions encoded in partition coefficients: an indirect estimation of hydrogen-bond parameters of polyfunctional solutes. Determination by centrifugal partition chromatography and factorization into steric/hydrophobic and polar components. J Chromatogr 556: 181–194Įl Tayar N, Tsai RS, Carrupt PA, Testa B (1992a) Octan-1-ol-water partition coefficients of zwitterionic α-amino acids. J Chromatogr 639: 287–294Įl Tayar N, Tsai RS, Vallat P, Altomare C, Testa B (1991) The use of CPC for assessing lipophilicity: a comparative evaluation. Biochem 29: 7133–7155ĭross K, Sonntag C, Mannhold R (1993) On the precise estimation of R M-values in reversedphase TLC including aspects of pH-dependence. Peptide Res 3: 75–80ĭill KA (1990) Dominant forces in protein folding. Quant Struct-Act Relat 12: 1–8Ĭowan R, Whittaker RG (1990) Hydrophobicity indices for amino acid residues as determined by high-performance liquid chromatography.

Collect Czech Chem Commun 56: 2030–2041Ĭocchi M, Johansson E (1993) Amino acids characterization by GRID and multivariate data analysis. J Theor Biol 99: 629–644Ĭhmelik J, Hudecek J, Putyera K, Makovicka J, Kalous V, Chmelikova J (1991) Characterization of the hydrophobic properties of amino acids on the basis of their partition and distribution coefficients in the 1-octanol-water system. Prog Phys Org Chem 18: 163–284Ĭharton M, Charton Bl (1982) The structural dependence of amino acid hydrophobicity parameters. Arch Biochem Biophys 161: 665–670Ĭharton M (1990) The quantitative description of amino acid, peptide, and protein properties and bioactivities.
J Chromatogr 373: 191–225īull HB, Breese K (1974) Surface tension of amino acid solutions: a hydrophobicity scale of the amino acid residues. Anal Biochem 193: 72–82īraumann T (1986) Determination of hydrophobic parameters by reversed-phase liquid chromatography: theory, experimental techniques and application in studies on quantitative structure-activity relationships.
Curr Op Struct Biol 2: 845–851īlack SD, Mould DR (1991) Development of hydrophobicity parameters to analyze proteins which bear post- or cotranslational modifications. Proteins Struct Funct Gen: 130–152īalaram P (1992) Non-standard amino acids in peptide design and protein engineering. Int J Biochem 2: 537–544Ībraham DJ, Leo A (1987) Extension of the fragment method to calculate amino acid zwitterion and side chain partition coefficients. Aboderin AA (1971) An empirical hydrophobicity scale for α-amino-acids and some of its applications.
- Blog
- Turnitin uf
- Obd-ii j2534 elm327 toad diagnostic software
- Dodonpachi resurrection black label xbox marketplace
- Contract killer 2 mod apk revdl
- Autodesk 3ds max 2012 material library download
- How to do edge blending in propresenter 6
- Are hydrophobic amino acids nonpolar
- Alien shooter pc
- Visual studio enterprise subscription azure
- Contoh proposal manajemen keuangan
- Clipp store download
- Regulador voltaje o supresor de picos
- How to add subtitles to video file permanently
